Chimeric Antigen Receptor (CAR) Overview

Introduction to CAR Cell Technology
Chimeric Antigen Receptor (CAR) cell technology represents a groundbreaking advancement in the field of immunotherapy, particularly in cancer treatment. This innovative approach involves genetically modifying a patient’s immune cells—most commonly T cells—to recognize and attack specific cancer cells more effectively. By engineering these cells to express synthetic receptors (CARs) on their surface, scientists can redirect the immune system to target tumor-associated antigens with high precision.
CAR cell therapy has shown remarkable success in treating certain types of blood cancers, such as leukemia and lymphoma, where traditional therapies may have limited effectiveness. The technology combines principles from molecular biology, genetic engineering, and immunology, making it a powerful example of personalized medicine. Despite its promising outcomes, challenges such as toxicity, high cost, and limited efficacy in solid tumors remain areas of active research.
Overall, CAR cell technology marks a significant step forward in the evolution of targeted therapies, offering new hope for patients with previously difficult-to-treat cancers.
Evolution of CAR Cell Technology
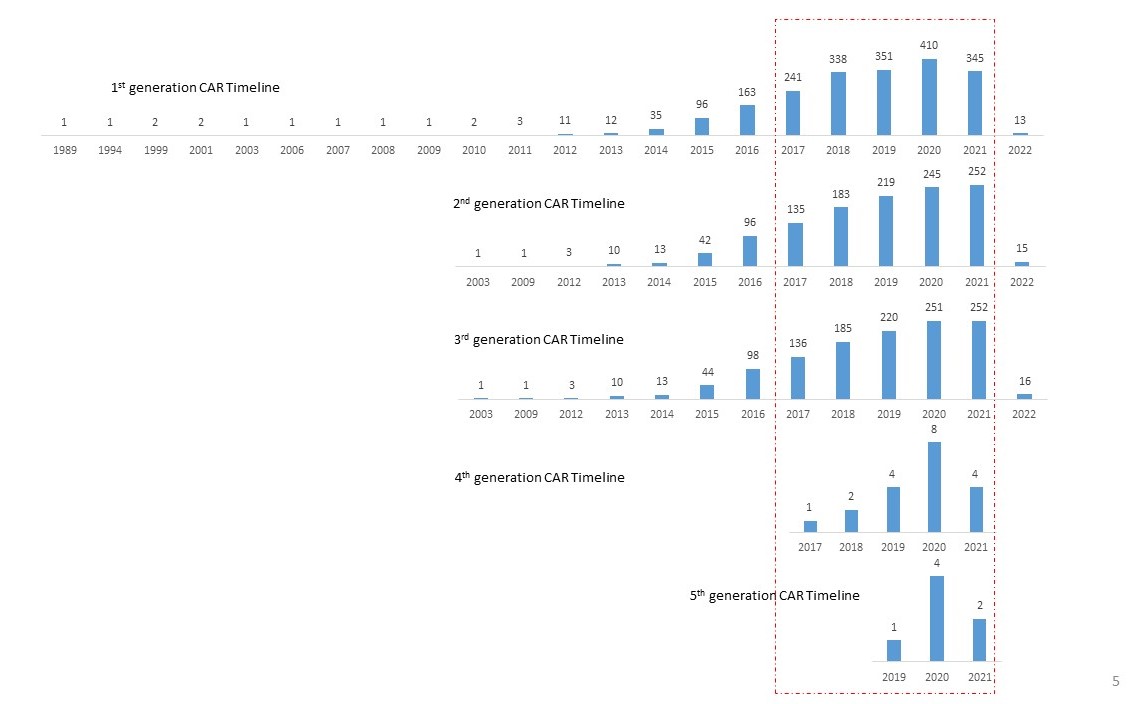
Among the 5572 patents, 3264 patents describe the CAR generation that is being used. Of these, the majority are from first generation(62%). The second and third generation appears in second place with (37%) each followed by fourth (0.6%) and fifth(0.25%).
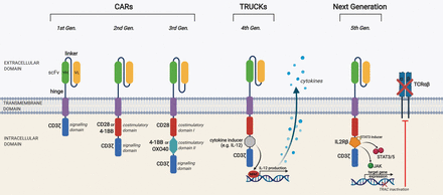
Evolution in CAR T-cell design
The process of modifying the intracellular domain to increase effectiveness led to the development of first-, second-, third-, and, recently, fourth-generation CARs.
- First-generation CARs consisted of only the TCR complex CD3-zeta chain domain and antigen recognition domains.
- Subsequently, second-generation CARs were developed, which incorporated costimulatory domains, such as CD28 or 4-1BB (CD137), thus increasing CAR-T cell survival and proliferation, ultimately leading to improved antitumor efficacy.
- Third-generation CARs combine the CD3-zeta domain with more than two costimulatory domains; nonetheless, its superiority over second-generation CARs has not yet been completely demonstrated.
- Finally, the fourth CAR generation, still in its early development, incorporates other genes to empower the antitumor activity of CAR-T cells
- Recently, other genetic modifications (i.e. receptor design or different vectors used for gene delivery) have been evaluated in pre-clinical (in vitro or in vivo biological testing) and clinical trial.
To date, there have been five main generations of CAR. The difference in each generation CAR mainly lies in the structure and functionality of the intracellular domain.
Problem:
Significant limitations have been noted for first, second and third -generation CARs, with less T-cell proliferation and inadequate release of cytokines.
Solution:
TRUCK, the fourth-generation CAR, stands for Tcells redirected for universal cytokine-mediated killing. This generation has resulted in a huge modification in the intracellular domain of the 2nd generation, i.e., the addition of cytokines such as interleukin 12 (IL-12), which activates natural killer (NK) cells in addition to other Tcells at the tumor site. IL-12 has the ability to cytolyse cancer cells that are not detected by CAR-T cells directly hence, IL-12 results in killing these cells by activating the innate immune response. The fifth generation of CAR has an additional modification of the binding site in the intracellular domain for the signal transducer and activator of transcription-3 (STAT3) transcription factor and another binding site for the interleukin-2 (IL-2) receptor. Both these modifications provide better T-cell activation, persistence and proliferation by cytokine-inducing Janus kinase (JAK–STAT3/5) signaling.
Why CAR cell Technology is important in therapy?
As revolutionary immuno-oncology changes to cancer treatment , cell therapies have attracted widespread attention with their high clinical remission rate in hematological cancers. Since 2018, cell therapies have accounted for more remissions than cancer vaccines , with chimeric antigen receptor (CAR) therapies leading the global cell therapy development race.
Genetic Modification of CAR-cells
Pending Issues
- The CAR cell success has thus far been limited to B cell malignancies. Extending this approach to other cancers will require the development of strategies based on understanding the obstacles posed by tumor heterogeneity and the tumor microenvironment that is emerging from sophisticated analytic tools and superior models.
- Among the clinical trials performed so far, the dose of administered CAR T cells varied substantially.
- Notably, there is no correlation between the infused number of CAR T cells and the clinical outcome including related toxicities.
- Along the same lines, CAR T cells can persist more than 6 years in patients and can lead to severe adverse events shortly after infusion as well as at later times.
- Finally, it must be stressed that CAR T cell therapy is still experimental and can be associated with significant risks for the patient. It becomes therefore of great importance to have a toxicity management plan in place and to identify biomarkers to predict common toxicities such as CRS.
About Effectual Services
Effectual Services is an award-winning Intellectual Property (IP) management advisory & consulting firm offering IP intelligence to Fortune 500 companies, law firms, research institutes and universities, and venture capital firms/PE firms, globally. Through research & intelligence we help our clients in taking critical business decisions backed with credible data sources, which in turn helps them achieve their organisational goals, foster innovation and achieve milestones within timelines while optimising costs.
We are one of the largest IP & business intelligence providers, globally serving clients for over a decade now. Our multidisciplinary teams of subject matter experts have deep knowledge of best practices across industries, are adept with benchmarking quality standards and use a combination of human and machine intellect to deliver quality projects. Having a global footprint in over 5 countries helps us to bridge boundaries and work seamlessly across multiple time zones, thus living to the core of our philosophy - Innovation is global, so are we !!!
Solutions Driving Innovation & Intelligence
Enabling Fortune 500's, R&D Giants, Law firms, Universities, Research institutes & SME's Around The Globe Gather Intelligence That
Protects and Nurtures Innovation Through a Team of 250+ Techno Legal Professionals.


